 This formulation has good agreement with experimental data and is comparable to established formulae for two-phase systems currently in use for lava, i.e., treatments based on the work by Einstein and Roscoe. This rigorously derived treatment reduces, in a limiting case, to a viscosity treatment of rigid spheres and fluid. In this paper, we examine the use of the three-phase treatment of Phan-Thien and Pham for a basaltic lava mixture. Three-phase treatments have been developed in other contexts such as in the study of suspensions and multiphase flow. Nevertheless, three-phase treatments have received less attention in the context of lava flows. 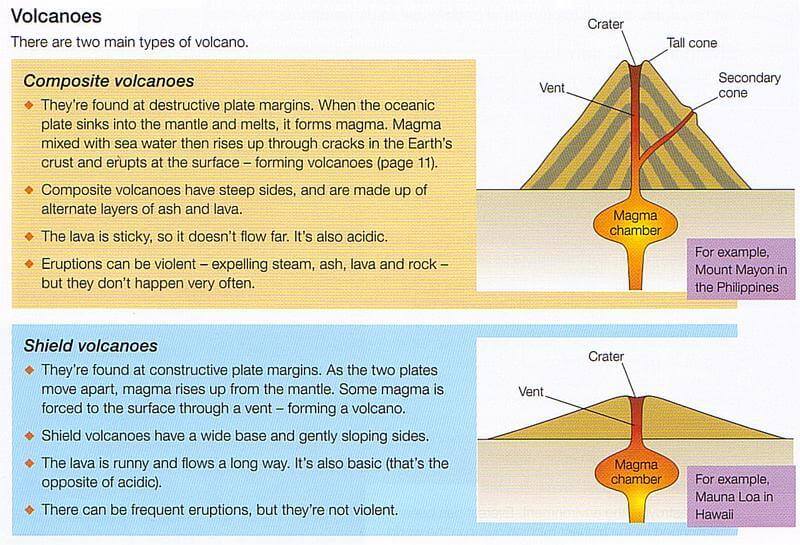
Dobran and Papale and Dobran, consider the three-phase viscosity for magma ascending a conduit during explosive Plinian eruptions. In addition, two-phase treatments have been applied to mixtures of fluid and crystals or fluid and bubbles. Single-phase treatments obtain fluid viscosity from composition, water content, and/or temperature. Many rheological treatments exist for one- and two-phase magma and/or lava mixtures. Derivation of viscosity is crucial in applications that seek to use field data and measurements such as lava flow temperature, crystallinity, and vesicularity for calculation of the bulk rheology of different suspensions, and/or to model lava flow dynamics. This mixture plays an influential role in determining the bulk rheology of the flow. The ability of the three-phase treatment to characterize the full range of melt-crystal-bubble mixture viscosities in both settings indicates the potential of this method in characterizing basaltic lava mixture viscosity.Ī basaltic lava is a three-phase mixture comprising a fluid component (basalt) plus voids (bubbles and/or space opening along shear lines) and solids (crystals, both phenocrysts and microphenocrysts). At Mauna Loa, the three-phase treatment provides a fit with the full range of field measured viscosities, giving three-phase mixture viscosities, upon eruption, of 110–140 Pa s (5% crystals, no bubble effect due to sheared vesicles) to 850–1400 Pa s (25–30% crystals, 40–60% spherical vesicles). This compares with a measured value for Etnean lava of 9400 ± 1500 Pa s. Application of a three-phase model yields mixture viscosities (45% crystals, 25–35% vesicles) in the range 5600–12,500 Pa s. % H 2O), and 135 ± 10 Pa s for less well-degassed (0.4 wt % H 2O), melt at 1080☌. At Etna, melt viscosities of 425 ± 30 Pa s are expected for well-degassed (0.1 w. We investigate existing one- and two-phase treatments using lava flow case studies from Mauna Loa (Hawaii) and Mount Etna (Italy) and compare these with a three-phase treatment that has not been applied previously to basaltic mixtures. However, three-phase treatments, allowing for the effects of coexisting crystallinity and vesicularity, are not well understood. While existing one-phase treatments allow melt phase viscosity to be assessed on the basis of composition, water content, and/or temperature, two-phase treatments constrain the effects of crystallinity or vesicularity on mixture viscosity. The VEI scale starts at 0 and has no upper limit, but the largest eruption in history was a magnitude 8.Lava flows comprise three-phase mixtures of melt, crystals, and bubbles. To better classify the size of volcanic eruptions, volcanologists developed the Volcanic Explosivity Index (VEI) to categorize eruptions. The more bubbles a magma develops, the higher the explosivity of the eruption. 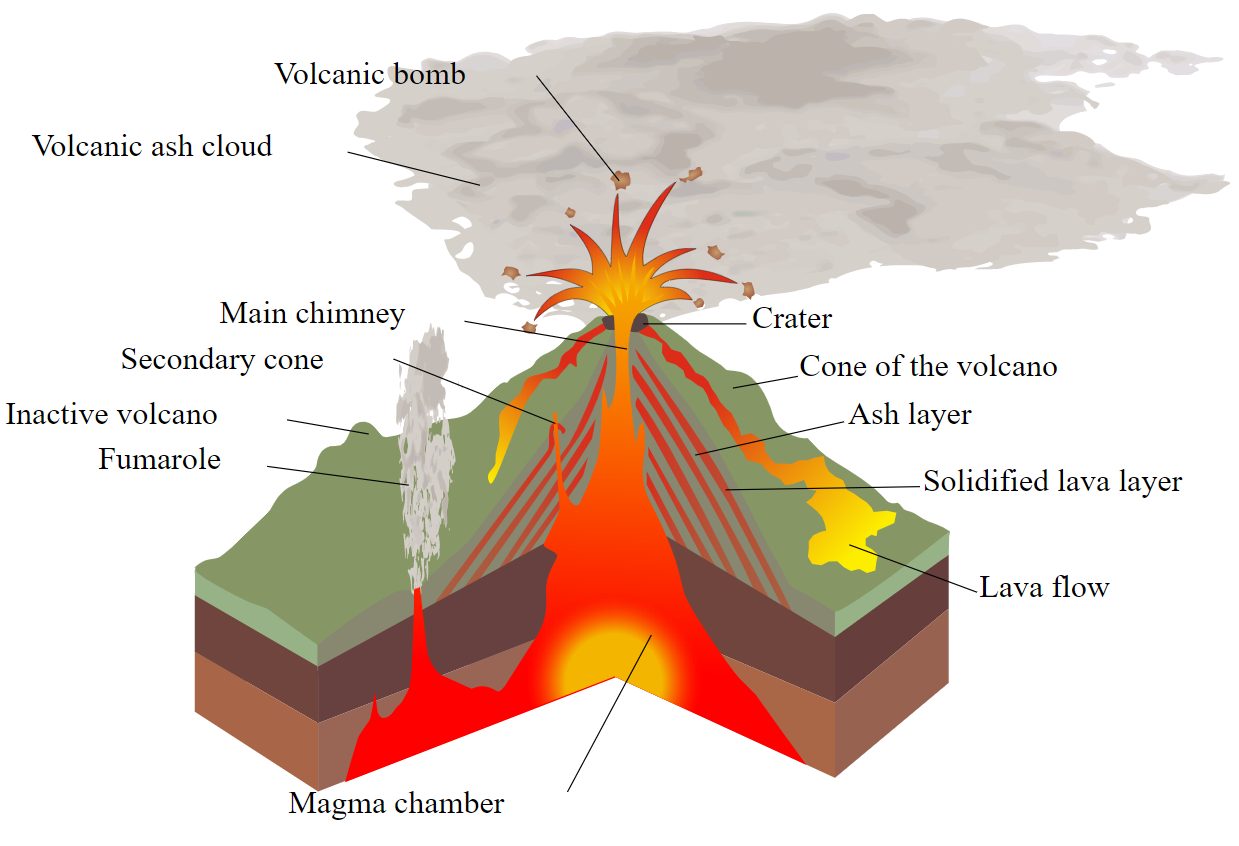 
If bubbles formed higher in the conduit, the magma would not fragment as much and the volcano would erupt more effusively.īubbles control a factor known in volcanology as explosivity. In the image to the left, bubbles form deep in the conduit of the volcano and rise to the surface, fragmenting the magma. A magma without bubbles will simply ooze to the surface. When a very bubbly magma reaches the surface, the bubbles pop and send shards of magma flying in every direction as pyroclasts. Sometimes, though, bubbles are not able to form. In some situations, bubbles form very easily. As magma rises towards the surface, though, the gases are no longer comfortable within the melt and come out of the magma to form bubbles. Deep within the earth as the magma resides in a magma chamber, these gases are comfortably mixed into the magma. Magmas contain many different gases such as H2O (water), CO2 (carbon dioxide), SO2 (sulfur dioxide), HCl (hydrogen chloride) and HF (hydrogen fluoride). Why do volcanoes behave in such different ways? Whether a volcano will erupt explosively or effusively is determined by the presence of bubbles. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
